Research
During early stages of embryogenesis, the developing central nervous system (CNS) appears strikingly conserved across vertebrate species. By the end of gestation, however, the vertebrate CNS has diversified dramatically, giving rise to an extraordinary range of species-specific morphologies and functions.
What drives the phylogenetic diversification of the vertebrate CNS?
Neural progenitors are the architects of the CNS, controlling both the types and numbers of neurons/glia that are generated. The phylogenetically divergent properties of neural progenitors are therefore key drivers that diversify the size, shape and function of the vertebrate CNS.
We aim to elucidate the unique properties of the human neural progenitor population, as well as their role in human development and disease.
Projects
Neural progenitor diversity
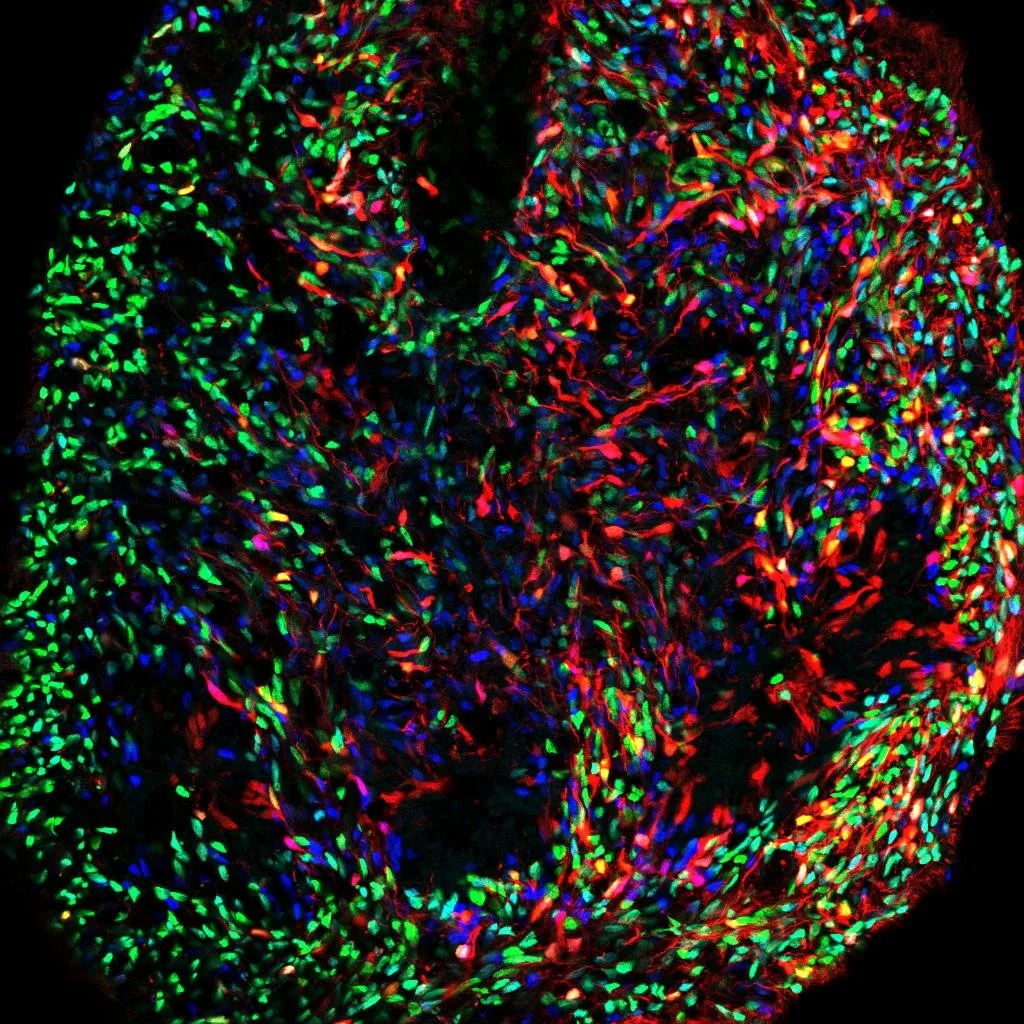
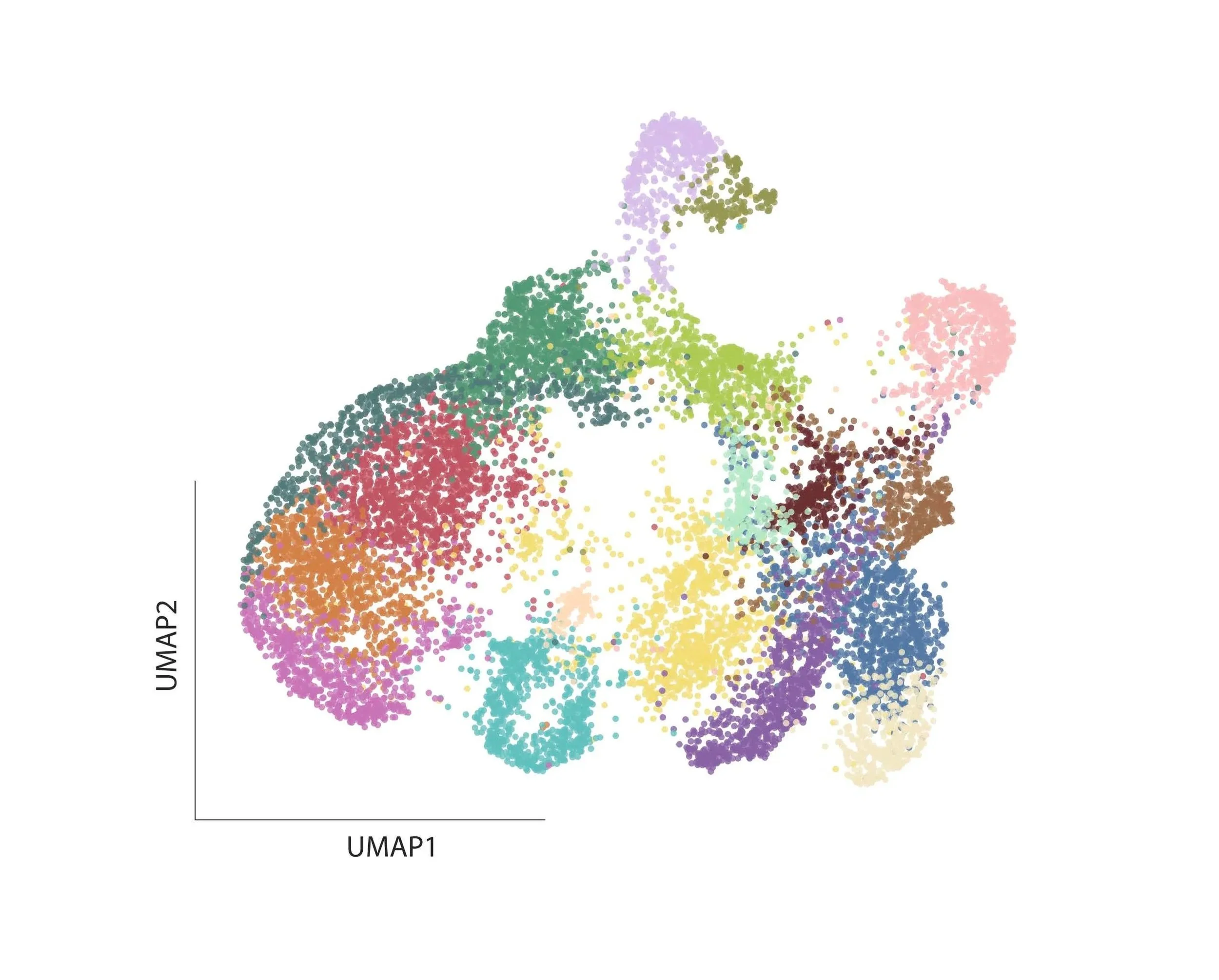
During human development, neural progenitors produce thousands of neuronal cell types, each defined as having distinct molecular, morphological, connectional, and/or electrophysiological properties. In contrast, neural progenitors themselves have traditionally been classified simply by marker gene expression, limiting our resolution as to their evident functional diversity.
To fill this gap in our understanding of neural development, we aim to utilize multiplexed, scalable genetic tools to systematically map the functional diversity of neural progenitors.
Human-specific gene regulation
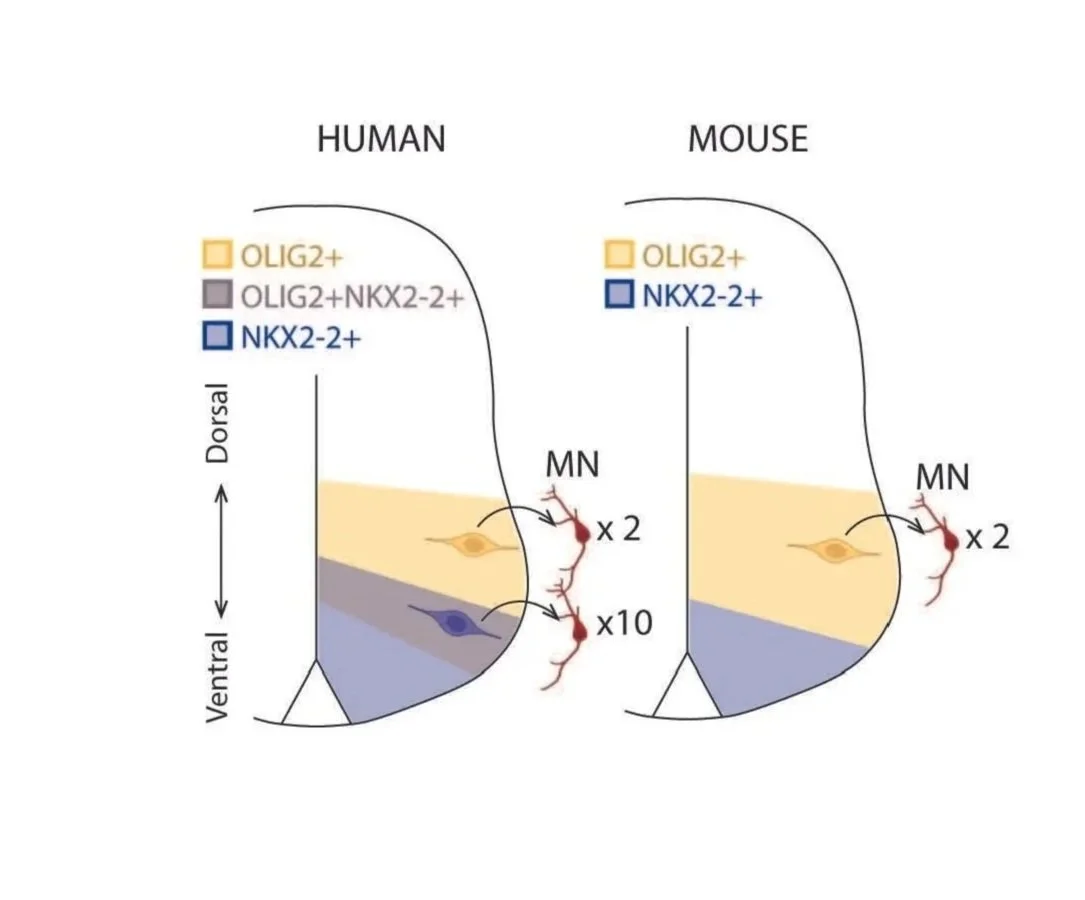
During development, cross-repressive interactions between key transcription factors pattern the neural epithelium into non-overlapping “domains” of neural progenitors. Interestingly, several of the boundaries between these domains are blurred in humans, giving rise to novel, functionally distinct progenitors.
We seek to understand the genetic basis of how these cross-repressive interactions are seemingly weakened in humans, as well as their downstream effects.
Human diseases
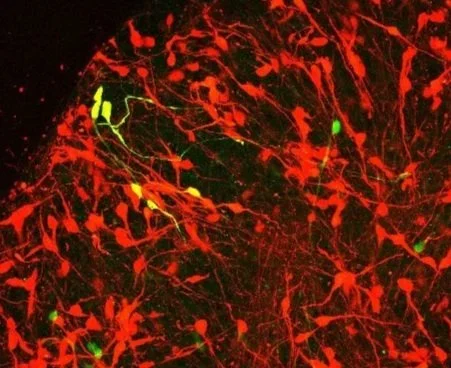
Most cell types in the human CNS are conserved among mammals, yet their susceptibility to disease differs across species. Importantly, many human diseases (such as ALS) do not occur naturally in most model organisms. We aim to leverage this phylogenetic variation in disease susceptibility to shed light on key pathogenic pathways.